See more ideas about atomic theory, physical science, teaching chemistry. In chemistry and physics, atomic theory is a scientific theory of the nature of matter, which states that matter is composed of discrete units called. Democritus’ atomic theory posited that all matter is made up small indestructible units he called atoms. Equivalently, one may define an atom to be an element that is minimal among the non-zero elements, or alternatively an element that covers the least element 0. Explore Kara Merancy's board 'atomic theory', followed by 146 people on Pinterest.
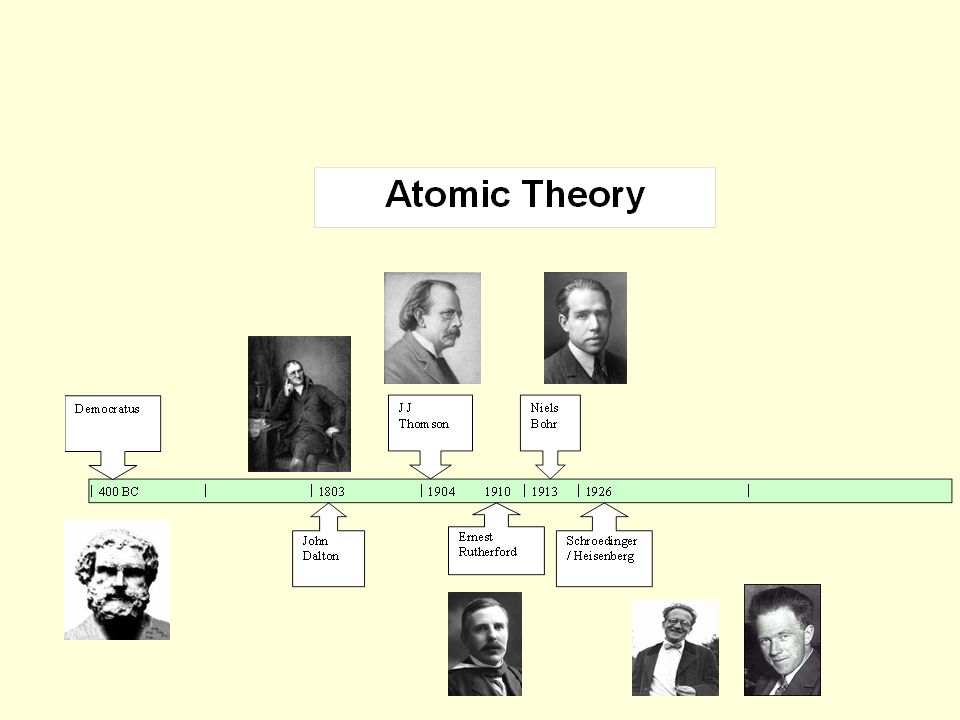
Choose from among the following scientists: Democritus, Rutherford, Thomson, Dalton, Bohr, Schrdinger, and Heisenberg. What is the Order of the atomic theory? Atom (order theory) In the mathematical field of order theory, an element a of a partially ordered set with least element 0 is an atom if 0 < a and there is no x such that 0 < x < a. In each box, write the name of the scientist(s) associated with the statement. Also he helped change the way people think of science. The atomic theory timeline shows the theories of the atom's construction over those years. What scientist contributed to the atomic theory? Robert Boyle's major contribution to the atomic theory was that he helped develop a definition of an element ( any substance that can be broken into 2 or more substances is not an element) and helped with " the death" of the four elements. The atomic theory has been developing and changing for over two thousand years. Because the Bohr Model is a modification of the earlier Rutherford Model, some people call Bohr's Model the Rutherford-Bohr Model. Who proposed the first model of an atom? Niels Bohr proposed the Bohr Model of the Atom in 1915. information atomic model analogy In the early 1800s, the English chemist John Dalton performed a number of experiments that eventually led to the acceptance of the idea of atoms. Z is the atomic number, the number of protons in the nucleus and the number electrons in the negative sphere. Info about Which Scientist Atomic Model Is Most Recent What is the earliest model of an atom? An early model of the atom, proposed by Rutherford after his discovery of the atomic nucleus, had a positive point charge, +Ze (the nucleus) at the center of a sphere of radius R with uniformly distributed negative charge -Ze.
